How To Draw Orbitals
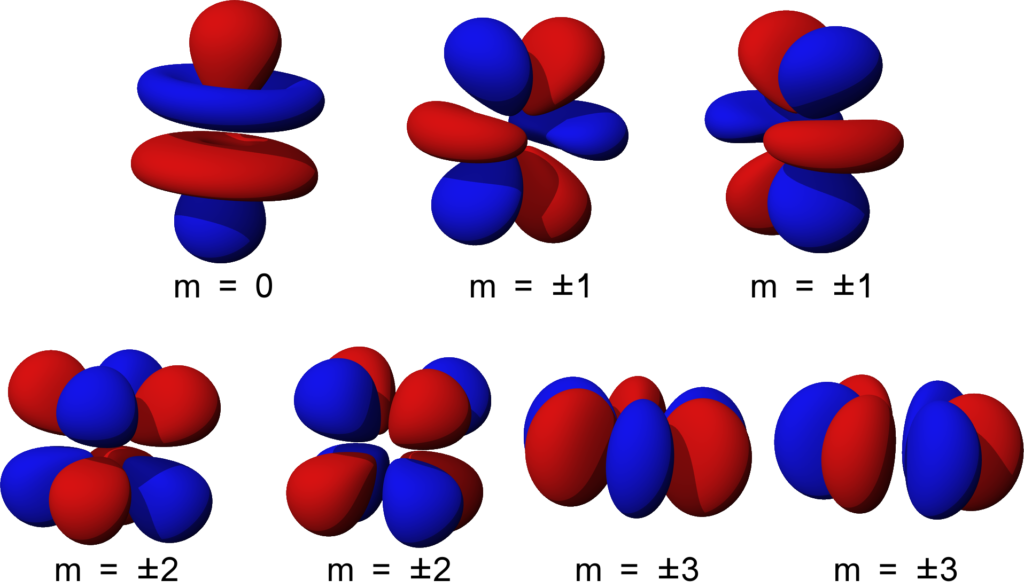
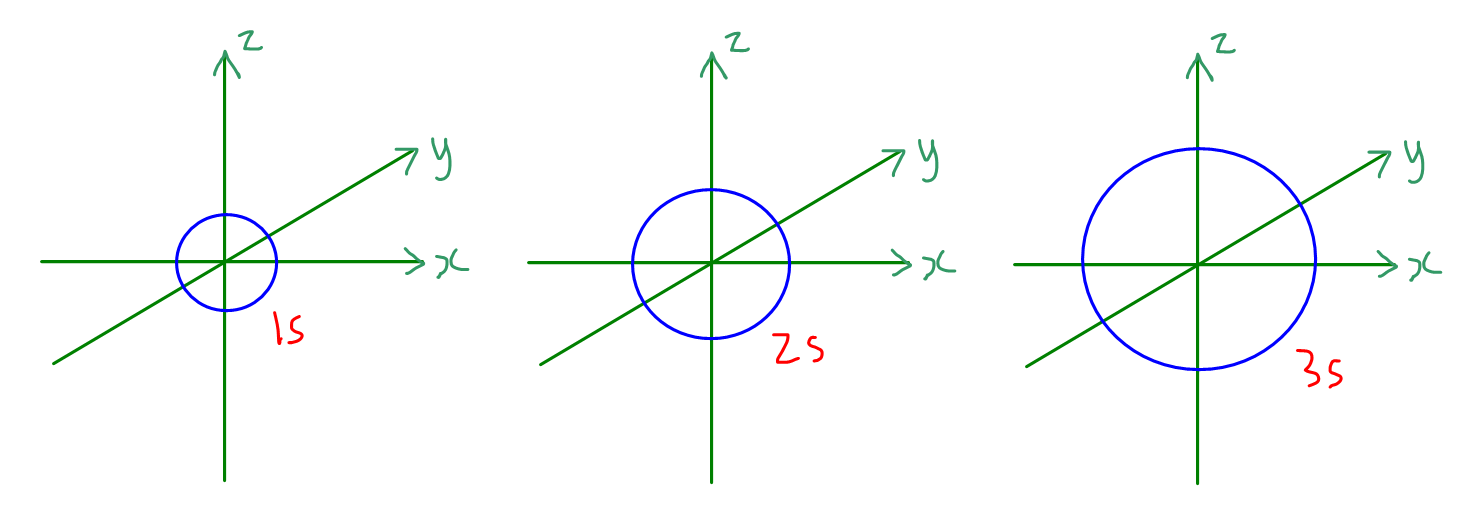
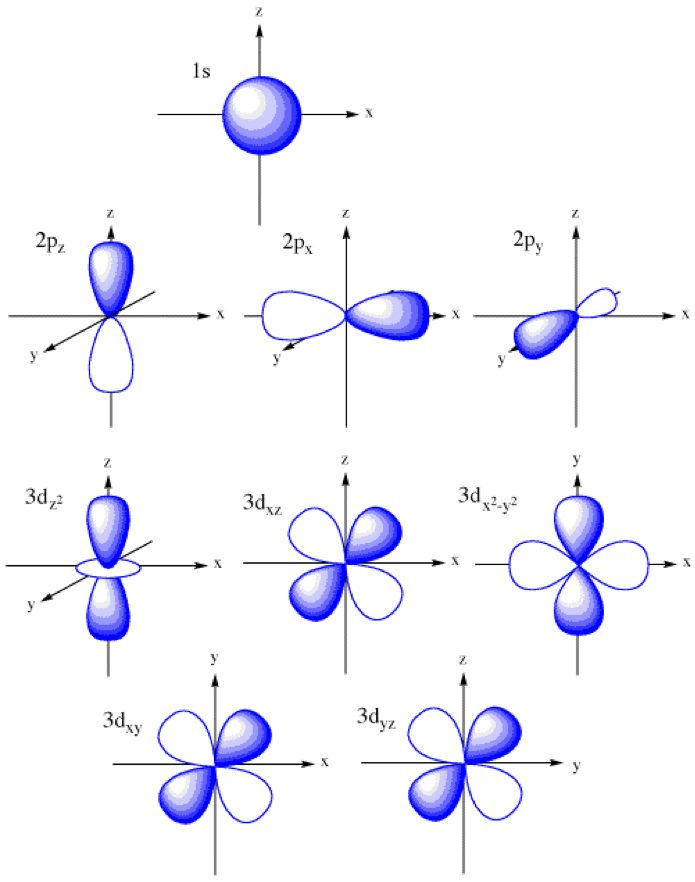
How To Draw Orbitals - 2.8k views 8 months ago physical chemistry. This article will explore the basics of how to draw each type of diagram, and important rules to follow in their construction. We're going to look at what orbitals are, what they represent, how electrons go in orbitals, the order electrons go in orbitals, and the shapes of. Web the basics of orbital diagrams. Are both of these stable molecules? 558k views 2 years ago chemistry. Web about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features nfl sunday ticket press copyright. Remember, arrows represent electrons, and lines or boxes are the orbitals. Ensure that these three rules are followed:. The aufbau principle, the pau. Orbitals with \(\ell = 2\) are d orbitals and have more complex shapes with at least two nodal surfaces. 558k views 2 years ago chemistry. For an s orbital, draw a circle; Are both of these stable molecules? For an f orbital, see below. Below are dot density diagrams, boundary surface diagrams, and a rotating image. Web this tutorial is about orbitals. A p orbital consists of two lobes of electron density on either side of the nucleus. L = 0, 1,., n − 1 l = 0, 1,., n − 1. A box, line, or circle, is drawn to represent each orbital in. Web 230313 how to draw shapes of orbitals. A box, line, or circle, is drawn to represent each orbital in the electron configuration. In an orbital diagram, an electron is represented by an arrow, while a box represents an atomic orbital. In it, the 2s orbitals and all three of the 2p orbitals hybridize to form four sp 3 orbitals,. Web typically, they only show the outermost electrons. Orbitals with \(\ell = 2\) are d orbitals and have more complex shapes with at least two nodal surfaces. A box, line, or circle, is drawn to represent each orbital in the electron configuration. As mentioned in the introduction, diagrams make use of horizontal lines which are filled with arrows to represent. The aufbau principle, the pau. For an f orbital, see below. Let's learn about the shapes of atomic orbitals in this video. This type of hybridization is required whenever an atom is surrounded by four groups of electrons. We classified the different orbital into shells and sub shells to distinguish them more easily. Web 230313 how to draw shapes of orbitals. The frontal lobes align themselves in the manner shown below. The aufbau principle, the pau. Let's learn about the shapes of atomic orbitals in this video. An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. Ml = −l,., −2, −1, 0, +1, +2,., l m l = − l,., − 2, − 1, 0, + 1, + 2,., l. An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. Web orbitals with \(\ell = 3\) are f orbitals, which are still more complex. Web this tutorial is. The frontal lobes align themselves in the manner shown below. As mentioned in the introduction, diagrams make use of horizontal lines which are filled with arrows to represent the spin direction of electrons. For an s orbital, draw a circle; This is also due to the history when they were discovered. Ensure that these three rules are followed:. Rank them in order of increasing stability. There are three different rules used for constructing an atomic orbital diagram. Web to draw orbitals, always start at the lowest energy level and build up. Web orbitals with \(\ell = 3\) are f orbitals, which are still more complex. An orbital is a space where a specific pair of electrons can be. Rank them in order of increasing stability. An orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. In two dimensions, we draw it as a circle. Web orbital diagrams are a visual way to show where the electrons are located within an atom. Web about press copyright contact us creators advertise developers. An s orbital is a sphere. Ml = −l,., −2, −1, 0, +1, +2,., l m l = − l,., − 2, − 1, 0, + 1, + 2,., l. The aufbau principle, the pau. This is also due to the history when they were discovered. Web this video goes over how to properly draw orbital diagrams for an element, after determining the electron configuration. Web to draw orbitals, always start at the lowest energy level and build up. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips. Are both of these stable molecules? N = 1, 2, 3,. Web draw the molecular orbitals for he 2 and he 2 +. We're going to look at what orbitals are, what they represent, how electrons go in orbitals, the order electrons go in orbitals, and the shapes of. Web about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features nfl sunday ticket press copyright. As mentioned in the introduction, diagrams make use of horizontal lines which are filled with arrows to represent the spin direction of electrons. In two dimensions, we draw it as a circle. N = 1, 2, 3,. There are three 2p orbitals, and they are at right angles to each other.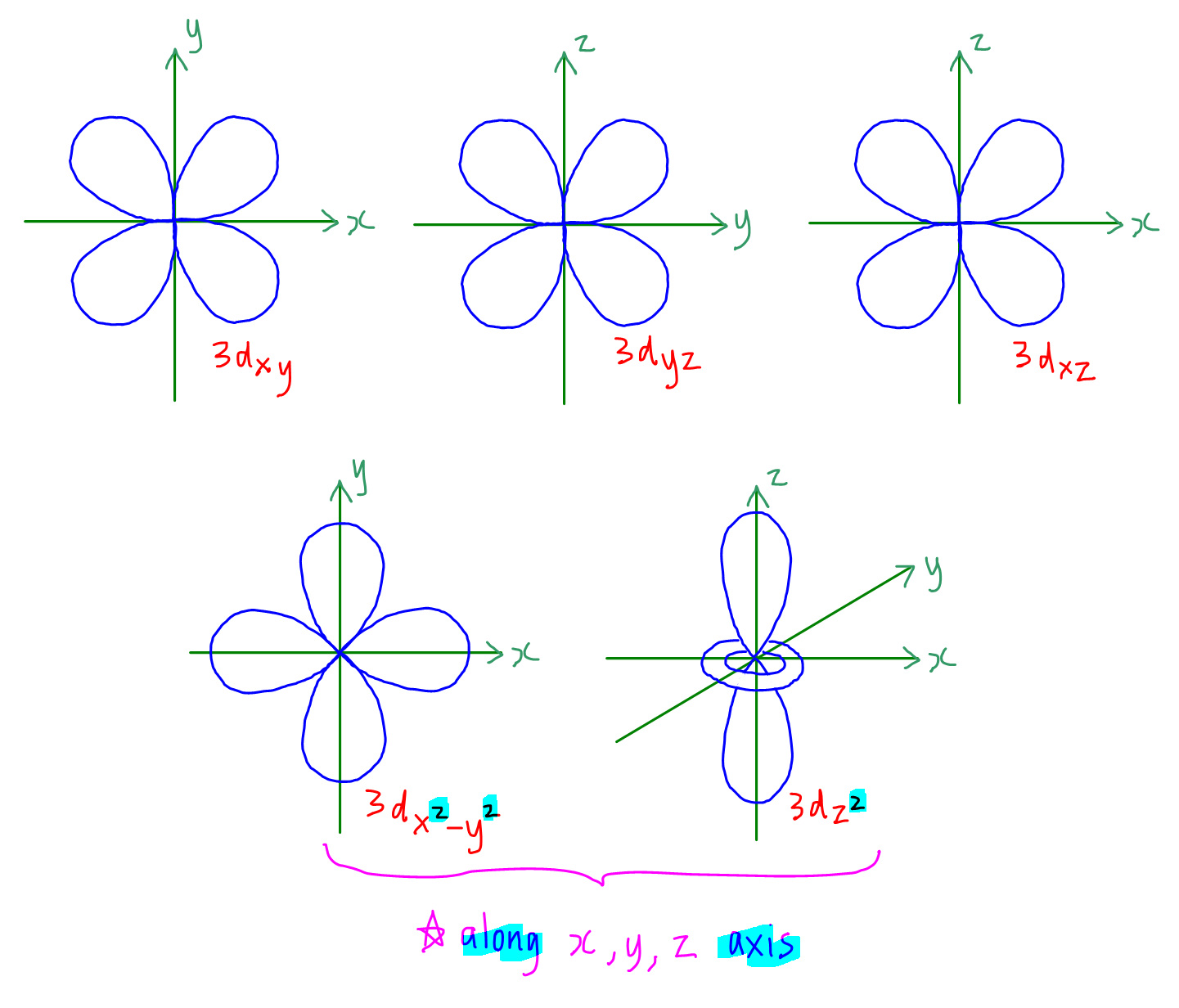
How to Draw Shapes of Orbitals

how to draw shapes of d orbitals elliottlyde
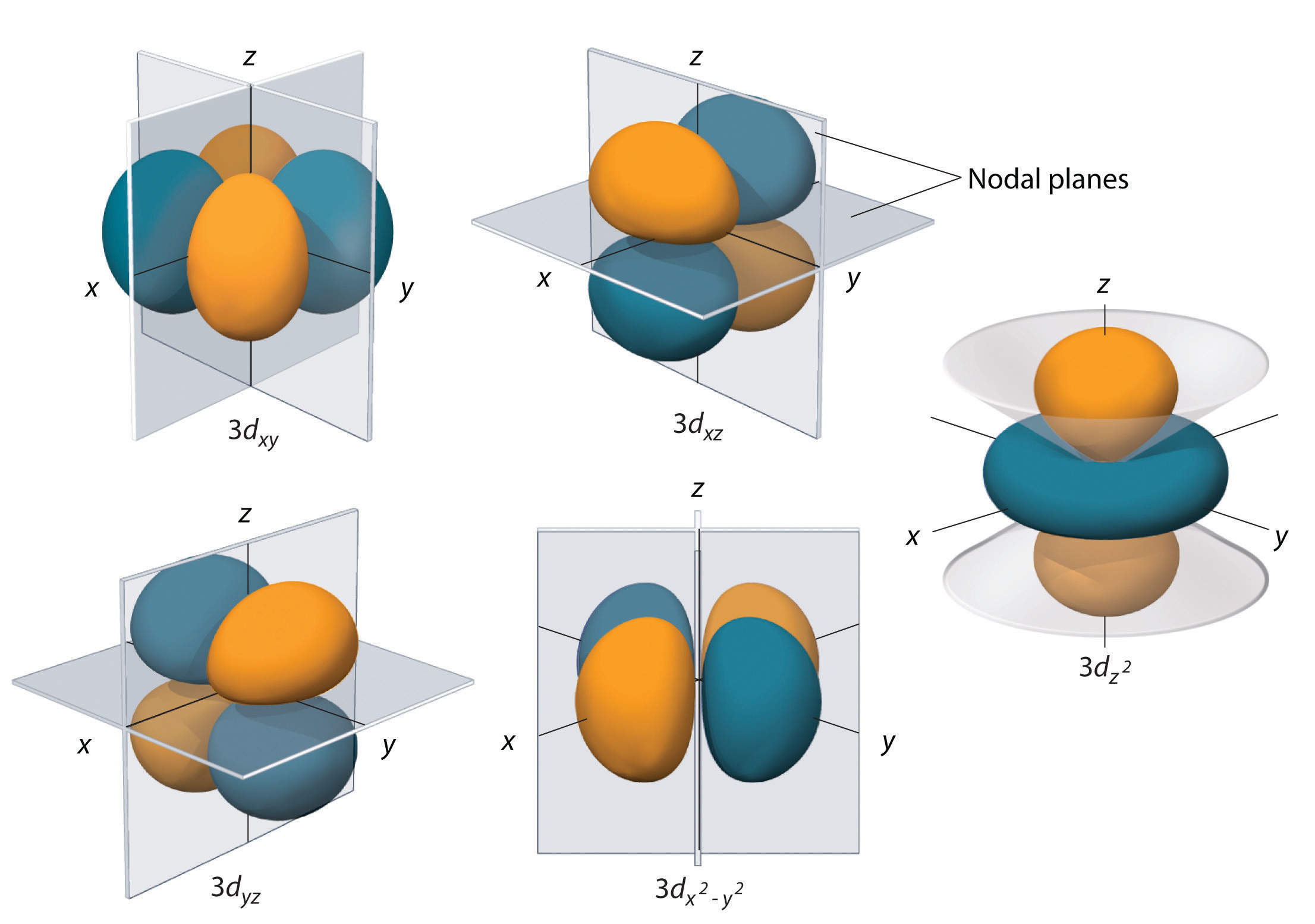
6.6 3D Representation of Orbitals Chemistry LibreTexts

12.1.5 Draw the shape of an s orbital and the shapes of the p x , p y
Shapes of Orbitals and their Types Chemistry Skills
How to Draw Shapes of Orbitals

How To Draw Orbitals Deepcontrol3

How do you draw s,p,d,f orbitals? Socratic

Shapes of Atomic Orbitals — Overview & Examples Expii
Atomic orbitals explained polizhuge
558K Views 2 Years Ago Chemistry.
We Classified The Different Orbital Into Shells And Sub Shells To Distinguish Them More Easily.
(Using The Aufau Principle To Order The Orbitals And Hence The Boxes, Lines Or Circles, As Shown Below)
This Type Of Hybridization Is Required Whenever An Atom Is Surrounded By Four Groups Of Electrons.
Related Post: