How Do You Draw Orbital Diagrams
How Do You Draw Orbital Diagrams - Try the following mo's on your own, and then check with the answers provided. Web to write electron configurations and draw orbital box diagrams, there are three rules that must be applied. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips. Remember, we can use the periodic table to help us. Four of them fill the 1s and 2s orbitals. Web an orbital is a space where a specific pair of electrons can be found. Web simon fraser university. This means that the first shell can hold 2 electrons. Intro to general chemistry 3h 51m. Atoms & elements 4h 15m. Typically, they only show the outermost electrons. Imagine shells around the nucleus, that get bigger and bigger. Web this chemistry video tutorial provides a basic introduction into orbital diagrams and electron configuration. Orbital diagrams must follow 3 rules: This is known as the aufbau principal. The second shell has 2 subshells: Web how to draw orbital diagrams. Web © 2024 google llc. It explains how to write the orbital diagram n. Carbon (atomic number 6) has six electrons. For example, the orbital diagram of li can be shown as: Web all orbitals with values of n > 1 n > 1 and ell = 0 e l l = 0 contain one or more nodes. Remember, we can use the periodic table to help us. The arrow shows a qualitative representation of increasing orbital energy. Carbon (atomic number. That's the number of valence electrons on. Answer • count the valence electrons on the molecule. Web © 2024 google llc. This is also due to the history when they were discovered. So we fill subshells in the order 1s 2s 2p because 1s is the lowest energy, 2s is higher energy, and 2p is highest energy. The second shell has 2 subshells: The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips. This video goes over how to properly draw orbital diagrams for an element, after determining the electron configuration. Web however, recall that the. Web when drawing orbital diagrams, we include empty boxes to depict any empty orbitals in the same subshell that we are filling. Web © 2024 google llc. Try the following mo's on your own, and then check with the answers provided. The orbital diagram or orbital notation simply represents the arrangement of electrons in different orbitals of an atom. Web. Web simon fraser university. Each box represents one orbital, and each arrow indicates one electron. How do you populate the electrons? We build electron configurations by filling the lowest energy orbitals first then filling progressively higher energy orbitals. Web drawing molecular orbital diagrams is one of the trickier concepts in chemistry. The second shell has 2 subshells: For example, the orbital diagram of li can be shown as: Web in orbitals diagrams, the orbitals are shown as boxes, and the electrons in them as arrows pointing up or down. Web this chemistry video tutorial provides a basic introduction into orbital diagrams and electron configuration. Carbon (atomic number 6) has six electrons. Web orbital diagrams are a visual way to show where the electrons are located within an atom. Web simon fraser university. The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips. Draw a long vertical arrow that points upward.. Web a p orbital which extends along the x axis is labeled a p x orbital. We classified the different orbital into shells and sub shells to distinguish them more easily. Draw a long vertical arrow that points upward. This article will explore the basics of how to draw each type of diagram, and important rules to follow in their. Web an orbital is a space where a specific pair of electrons can be found. Electron orbital diagrams and written configurations tell you which orbitals are filled and which are partially filled for any atom. Write out the electron configuration to determine which orbitals are filled. We classified the different orbital into shells and sub shells to distinguish them more easily. The aufbau principle, the pau. Web an orbital diagram, like those shown above, is a visual way to reconstruct the electron configuration by showing each of the separate orbitals and the spins on the electrons. Web every subshell has a # of orbits s/p/d/f that can each hold 2 electrons each (one has the opposite spin of the other). Remember, we can use the periodic table to help us. Each box represents one orbital, and each arrow indicates one electron. Four of them fill the 1s and 2s orbitals. Try the following mo's on your own, and then check with the answers provided. Web © 2024 google llc. Web this chemistry video tutorial provides a basic introduction into orbital diagrams and electron configuration. For example, the orbital diagram of li can be shown as: The number of valence electrons impacts on their chemical properties, and the specific ordering and properties of the orbitals are important in physics, so many students have to get to grips. The remaining two electrons occupy the 2p subshell.
Molecular Orbitals Introductory Chemistry
3.7 Electron Arrangement The Quantum Model Chemistry LibreTexts

How To Draw Orbitals Deepcontrol3
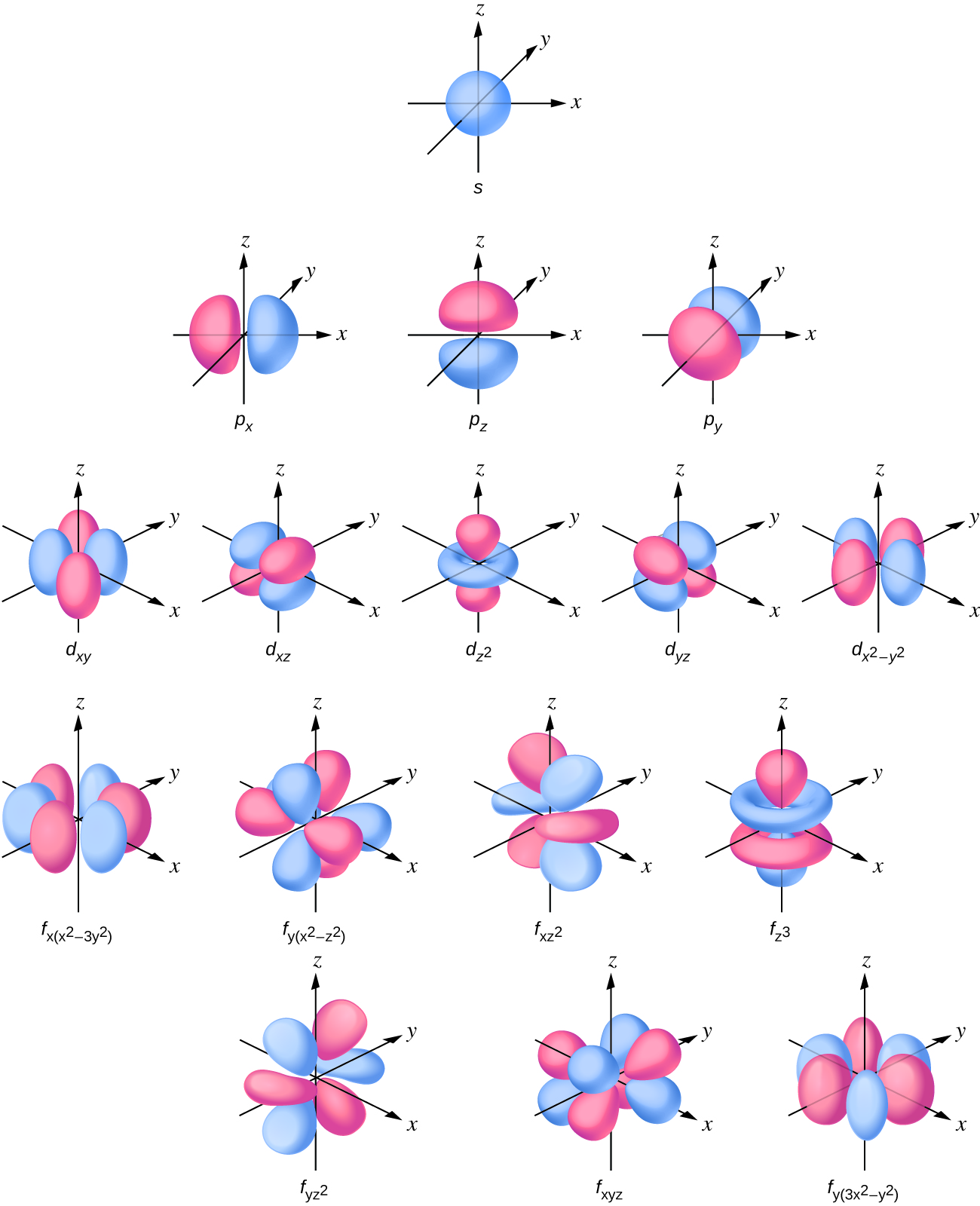
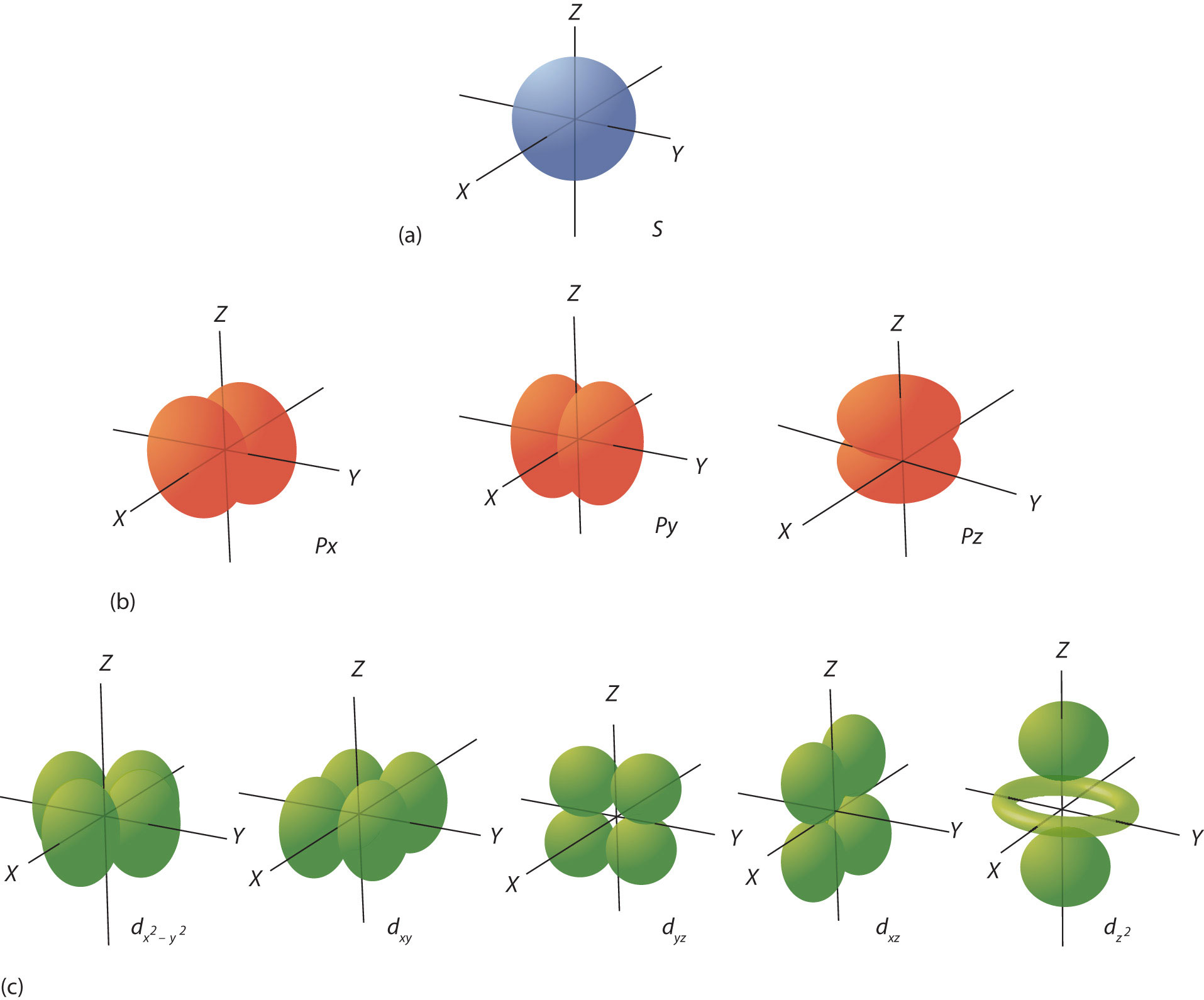
6.6 The Shapes of Atomic Orbitals Chemistry LibreTexts

O2 Molecular Orbital Diagrams 101 Diagrams
Radial and Angular Parts of Atomic Orbitals Chemistry LibreTexts

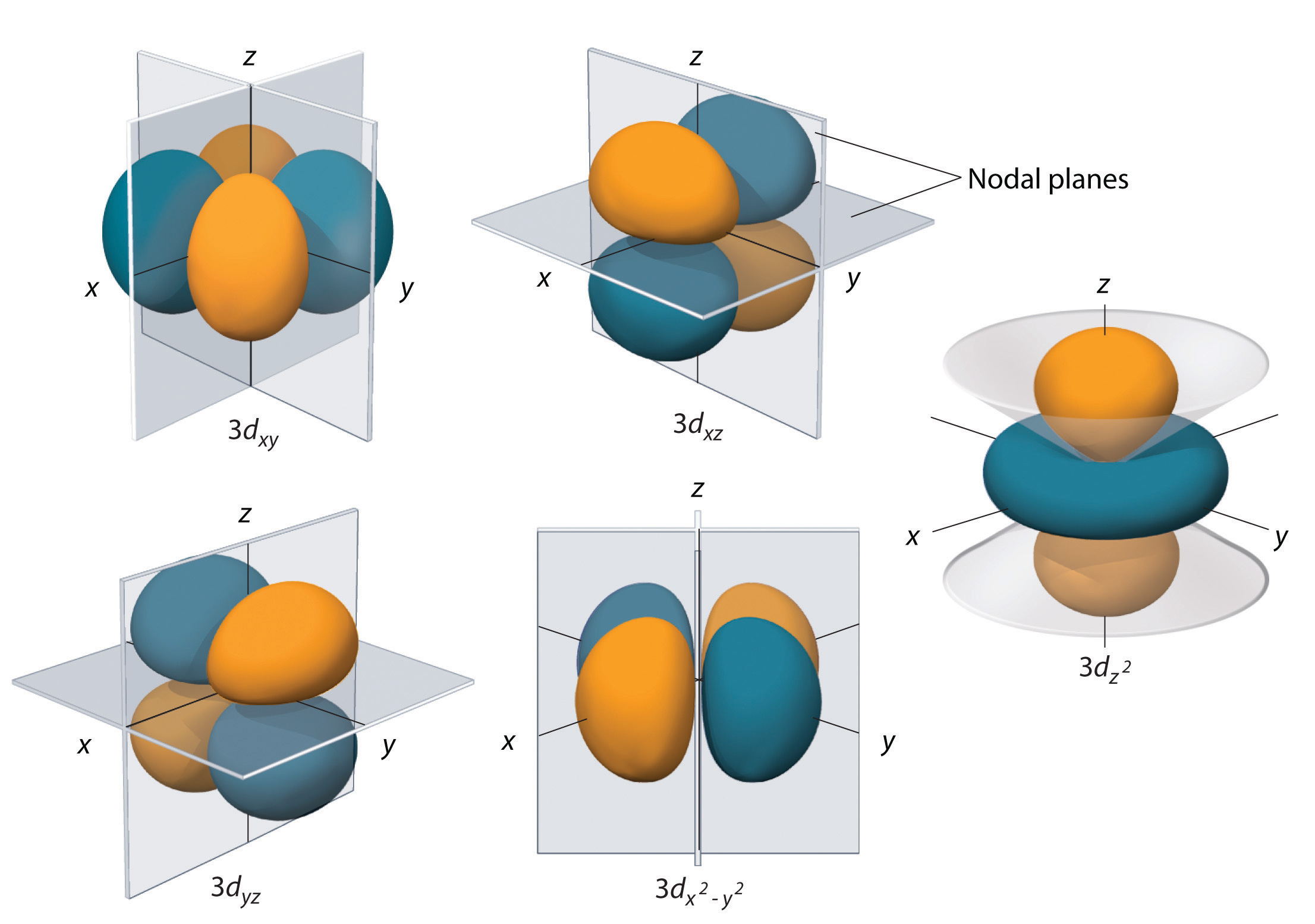
how to draw shapes of d orbitals elliottlyde

How To Do Orbital Diagrams exatin.info

how to draw molecular orbital diagram for heteronuclear molecules

How To Draw Orbitals Deepcontrol3
This Is Known As The Aufbau Principal.
Orbitals With ℓ = 2 ℓ = 2 Are D Orbitals And Have More Complex Shapes With At Least Two Nodal Surfaces.
The Second Shell Has 2 Subshells:
Web When Drawing Orbital Diagrams, We Include Empty Boxes To Depict Any Empty Orbitals In The Same Subshell That We Are Filling.
Related Post: