Drawing Resonance Structures
Drawing Resonance Structures - There is one pi bond in a double bond and two pi bonds in a triple bond.) never break a single bond (also known as a sigma bond). Work in one region at a time. Web when drawing resonance structures, here are a few things you want to keep in mind: Web some molecules have two or more chemically equivalent lewis electron structures, called resonance structures. Web this organic chemistry video tutorial provides a basic introduction into drawing resonance structures. Only move the electrons ( never move the atoms). Molecules with only single bonds never show resonance. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. Use the concept of resonance to explain structural features of molecules and ions. Determine the relative stability of resonance structures using a set of rules. Resonance only occurs when a molecule has at least one double bond. Determine the relative stability of resonance structures using a set of rules. Resonance structures, hybridization of orbitals, and localized and delocalized electron density. It explains how to identify the major resonance contributor as well as the minor. Web when drawing resonance structures, here are a few things you. All right, let's look at an application of the acetate anion here, and the resonance structures that we can draw. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. It explains how to identify the major resonance contributor as well as the minor. Resonance structures, hybridization of. Use the concept of resonance to explain structural features of molecules and ions. Only move the electrons ( never move the atoms). Web draw the resonance structures of molecules or ions that exhibit delocalization. Resonance only occurs when a molecule has at least one double bond. Web in the case that more than one reasonable (plausible) lewis structure can be. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. Resonance structures, hybridization of orbitals, and localized and delocalized electron density. Resonance only occurs when a molecule has at least one double bond. We have learned that lewis structure is a straightforward representation of valence shell electrons in. Web when drawing resonance structures, here are a few things you want to keep in mind: Resonance only occurs when a molecule has at least one double bond. There is one pi bond in a double bond and two pi bonds in a triple bond.) never break a single bond (also known as a sigma bond). Determine the relative stability. Web resonance occurs when we can draw two or more legitimate lewis structures for the same molecule. Web draw the resonance structures of molecules or ions that exhibit delocalization. There is one pi bond in a double bond and two pi bonds in a triple bond.) never break a single bond (also known as a sigma bond). Resonance structures, hybridization. Web some molecules have two or more chemically equivalent lewis electron structures, called resonance structures. Resonance only occurs when a molecule has at least one double bond. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. Web resonance occurs when we can draw two or more legitimate. We have learned that lewis structure is a straightforward representation of valence shell electrons in an atom, ion, or molecule. Web draw the resonance structures of molecules or ions that exhibit delocalization. Work in one region at a time. Web some molecules have two or more chemically equivalent lewis electron structures, called resonance structures. There is one pi bond in. Web in the case that more than one reasonable (plausible) lewis structure can be drawn for a species, these structures are called resonance structures or resonance contributors. Work in one region at a time. Only move the electrons ( never move the atoms). This includes lone pair electrons and pi bonds (remember: Web when drawing resonance structures, here are a. Work in one region at a time. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. Web in the case that more than one reasonable (plausible) lewis structure can be drawn for a species, these structures are called resonance structures or resonance contributors. Want to join the. Web some molecules have two or more chemically equivalent lewis electron structures, called resonance structures. Resonance is a mental exercise and method within the valence bond theory of bonding that describes the delocalization of electrons within molecules. Work in one region at a time. Resonance only occurs when a molecule has at least one double bond. Want to join the conversation? Use the concept of resonance to explain structural features of molecules and ions. Only move the electrons ( never move the atoms). Web in the case that more than one reasonable (plausible) lewis structure can be drawn for a species, these structures are called resonance structures or resonance contributors. Web resonance occurs when we can draw two or more legitimate lewis structures for the same molecule. Determine the relative stability of resonance structures using a set of rules. This includes lone pair electrons and pi bonds (remember: It explains how to identify the major resonance contributor as well as the minor. Web when drawing resonance structures, here are a few things you want to keep in mind: All right, let's look at an application of the acetate anion here, and the resonance structures that we can draw. Resonance structures, hybridization of orbitals, and localized and delocalized electron density. Molecules with only single bonds never show resonance.
3 manières de dessiner une représentation de Lewis

Resonance Structures, Basic Introduction How To Draw The Resonance

How to draw resonance structures YouTube
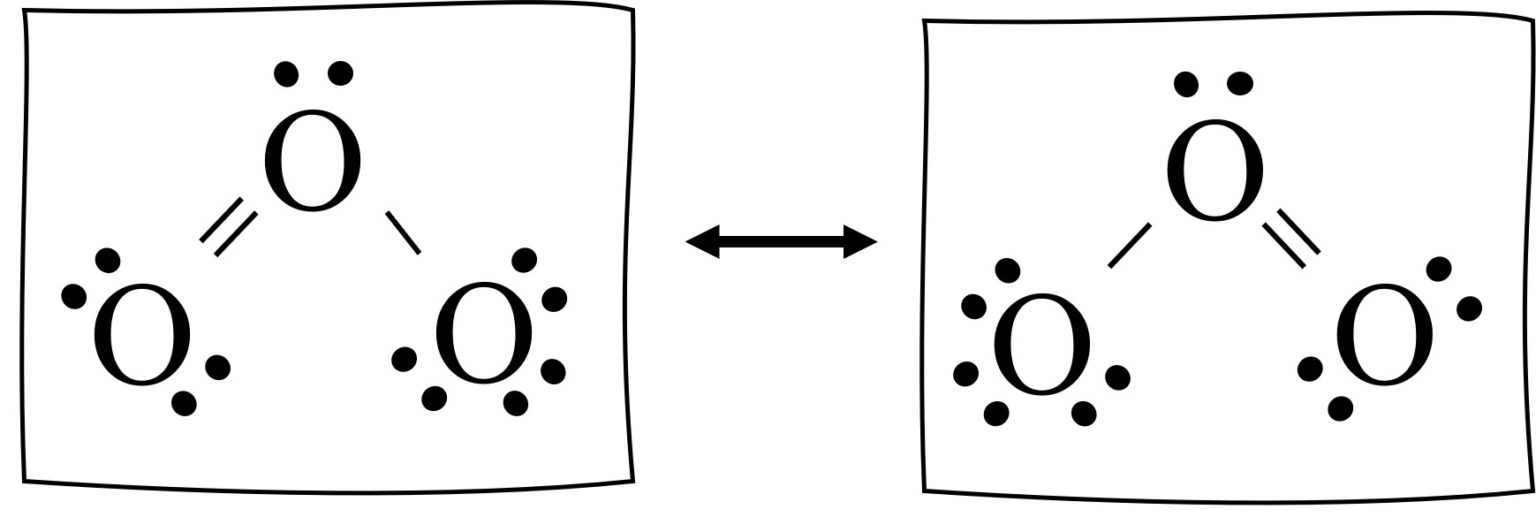
Resonance Structures Easy Hard Science

How to Draw Resonance Contributors MCC Organic Chemistry

Resonance Structures YouTube
[Solved] How to draw resonance structures of a complicated molecule
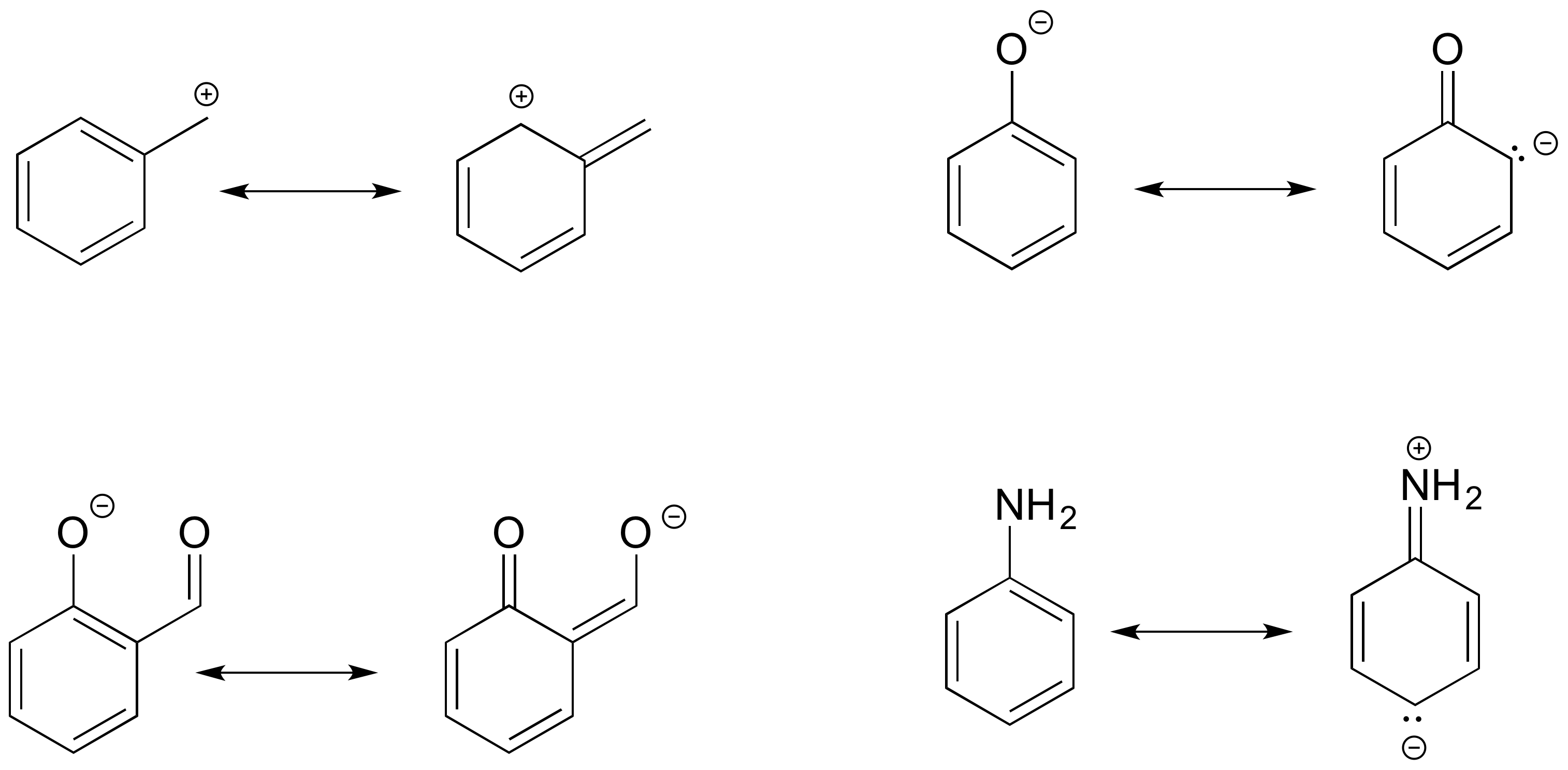
1.8 Drawing Resonance Forms Chemistry LibreTexts
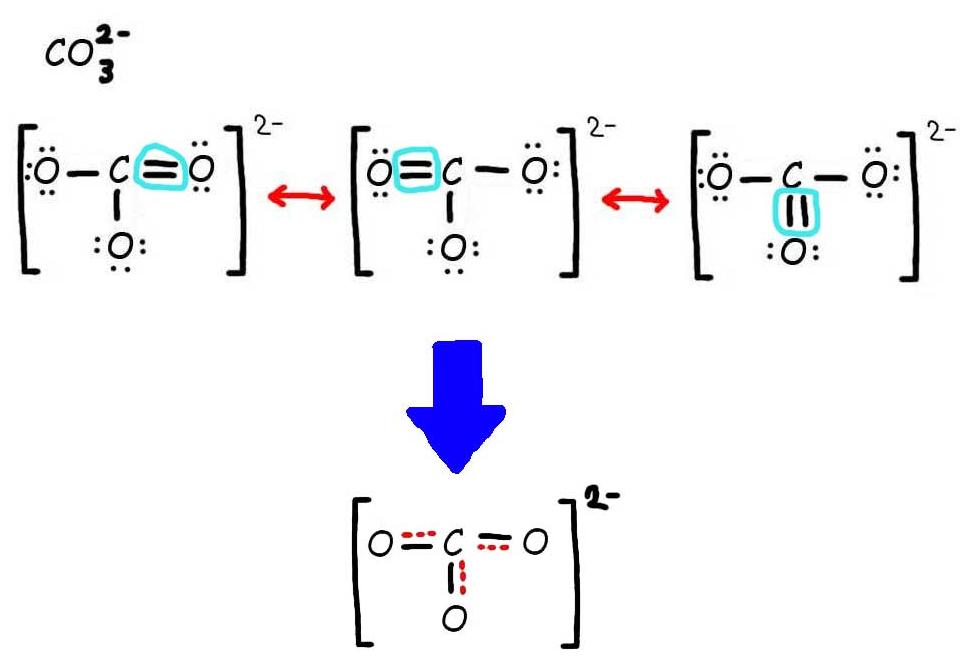
Resonance Chemwiki
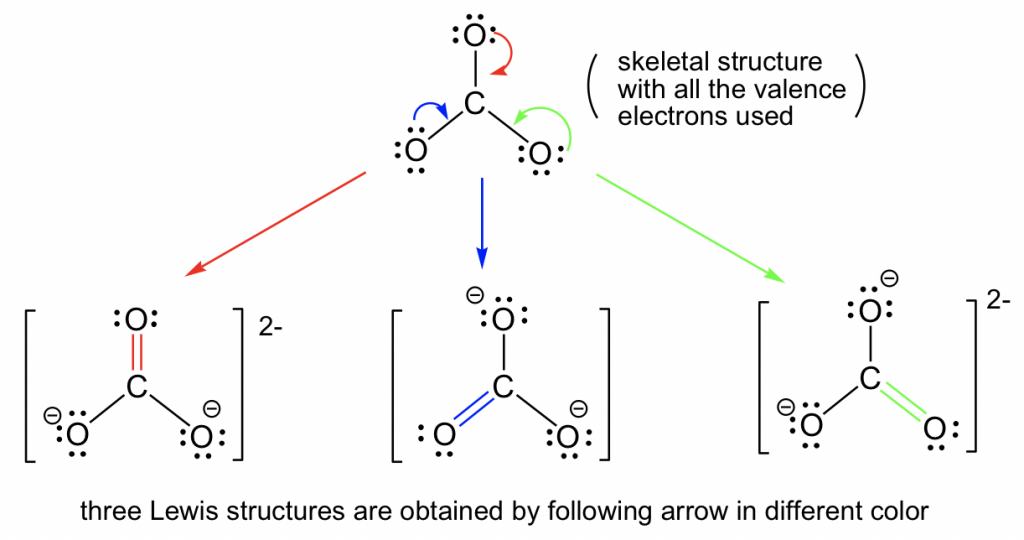
1.3 Resonance Structures Chemistry LibreTexts
Web This Organic Chemistry Video Tutorial Provides A Basic Introduction Into Drawing Resonance Structures.
Web Draw The Resonance Structures Of Molecules Or Ions That Exhibit Delocalization.
We Have Learned That Lewis Structure Is A Straightforward Representation Of Valence Shell Electrons In An Atom, Ion, Or Molecule.
There Is One Pi Bond In A Double Bond And Two Pi Bonds In A Triple Bond.) Never Break A Single Bond (Also Known As A Sigma Bond).
Related Post: