Draw The Lewis Structure Of Clo3
Draw The Lewis Structure Of Clo3 - Count the total number of valence electrons in the molecule. 182k views 12 years ago every video. It will hold more than 8 electrons. 73k views 3 years ago new ap & general chemistry video playlist. Subscribe:in this video, you will learn how to draw the lewis structure for. Learn how to draw the lewis structure of the chlorate ion and understand its stability. Put one electron pair in each bond4. The resonance structures of the cyanate ion are explored in example 9.8. Put least electronegative atom in centre3. #2 next, indicate lone pairs on the atoms. 182k views 12 years ago every video. Assign formal charges to each atom. Web 405k views 10 years ago. 70 more lewis dot structures. You might think you've got the correct. Web chemistry learning made easy! Subscribe:in this video, you will learn how to draw the lewis structure for. Web so, the lewis structure of clo3+ is: Put least electronegative atom in centre3. Web the cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. The resonance structures of the cyanate ion are explored in example 9.8. 182k views 12 years ago every video. 73k views 3 years ago new ap & general chemistry video playlist. Subscribe:in this video, you will learn how to draw the lewis structure for. 70 more lewis dot structures. Chlorine does not follow the octet rule. Subscribe:in this video, you will learn how to draw the lewis structure for. You might think you've got the correct. 73k views 3 years ago new ap & general chemistry video playlist. #2 next, indicate lone pairs on the atoms. Learn how to draw the lewis structure of the chlorate ion and understand its stability. It will hold more than 8 electrons. Web the cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. Web 405k views 10 years ago. For this structure, give each atom an octet and do not include a formal charge. Web the cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. Web chemistry learning made easy! Web 405k views 10 years ago. The resonance structures of the cyanate ion are explored in example 9.8. Web so, the lewis structure of clo3+ is: You might think you've got the correct. Subscribe:in this video, you will learn how to draw the lewis structure for. Web lewis dot of the chlorate ion. Learn how to draw the lewis structure of the chlorate ion and understand its stability. It will hold more than 8 electrons. The resonance structures of the cyanate ion are explored in example 9.8. #1 draw a rough sketch of the structure. Web the cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. For this structure, give each atom an octet and do not include a formal charge. Subscribe:in this video, you will learn how to draw. 70 more lewis dot structures. The resonance structures of the cyanate ion are explored in example 9.8. 182k views 12 years ago every video. It will hold more than 8 electrons. You might think you've got the correct. Web lewis dot of the chlorate ion. Draw a lewis structure that obeys the octet rule for each of the following ions. Chlorine does not follow the octet rule. It will hold more than 8 electrons. #2 next, indicate lone pairs on the atoms. Chlorine does not follow the octet rule. Subscribe:in this video, you will learn how to draw the lewis structure for. #2 next, indicate lone pairs on the atoms. Learn how to draw the lewis structure of the chlorate ion and understand its stability. Count the total number of valence electrons in the molecule. Draw a lewis structure that obeys the octet rule for each of the following ions. #1 draw a rough sketch of the structure. Put least electronegative atom in centre3. You might think you've got the correct. Web lewis dot of the chlorate ion. Web chemistry learning made easy! Assign formal charges to each atom. Web so, the lewis structure of clo3+ is: 73k views 3 years ago new ap & general chemistry video playlist. Web the cyanate ion is stable, while the fulminate ion is unstable and forms explosive compounds. 182k views 12 years ago every video.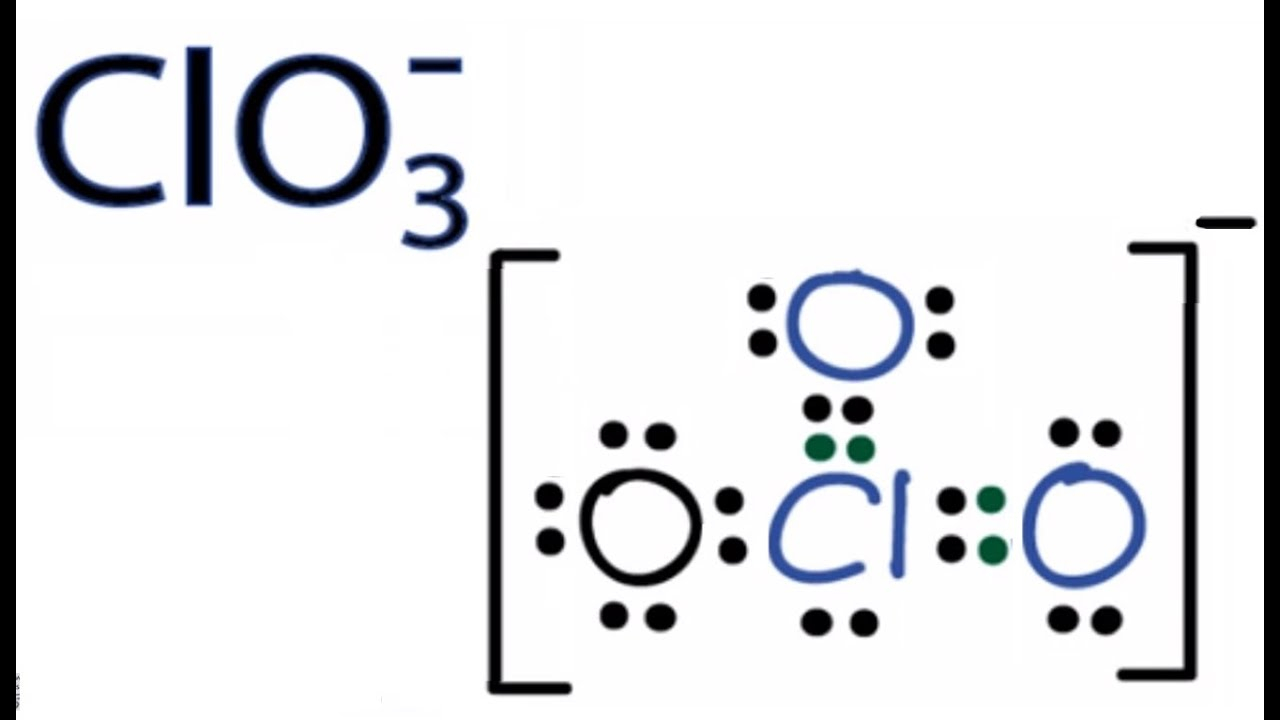
Electron Dot Diagram For Chlorine exatin.info
ClO3 Lewis Structure Chlorate Ion YouTube

ClO3 Lewis Structure (Chlorate Ion) YouTube
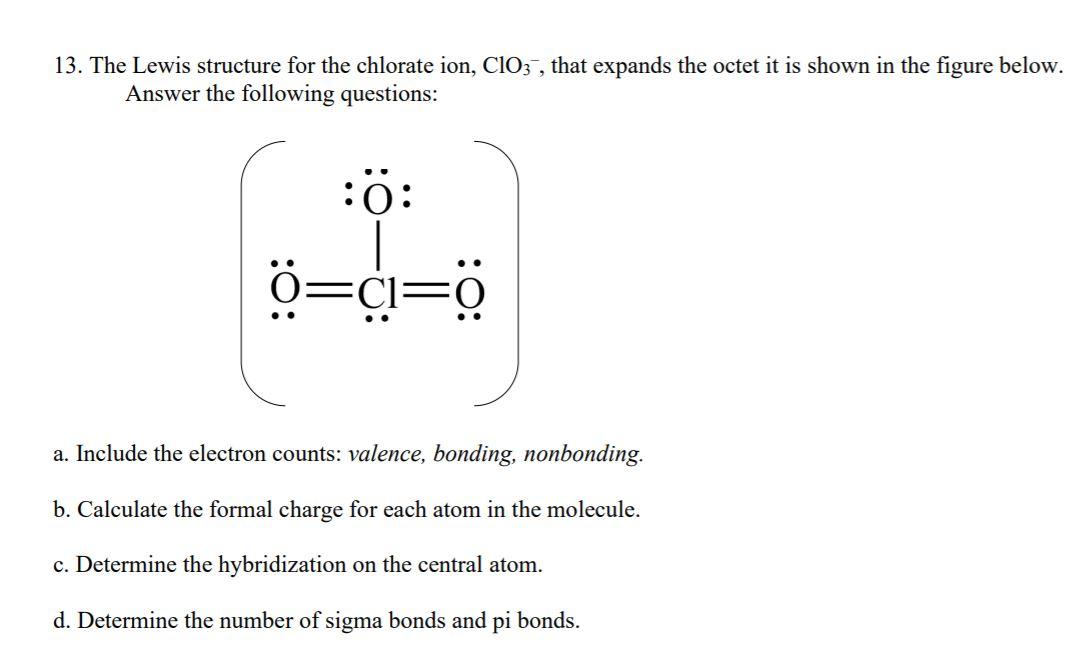
Solved 13. The Lewis structure for the chlorate ion, ClO3 ,
Clo3 Lewis Structure
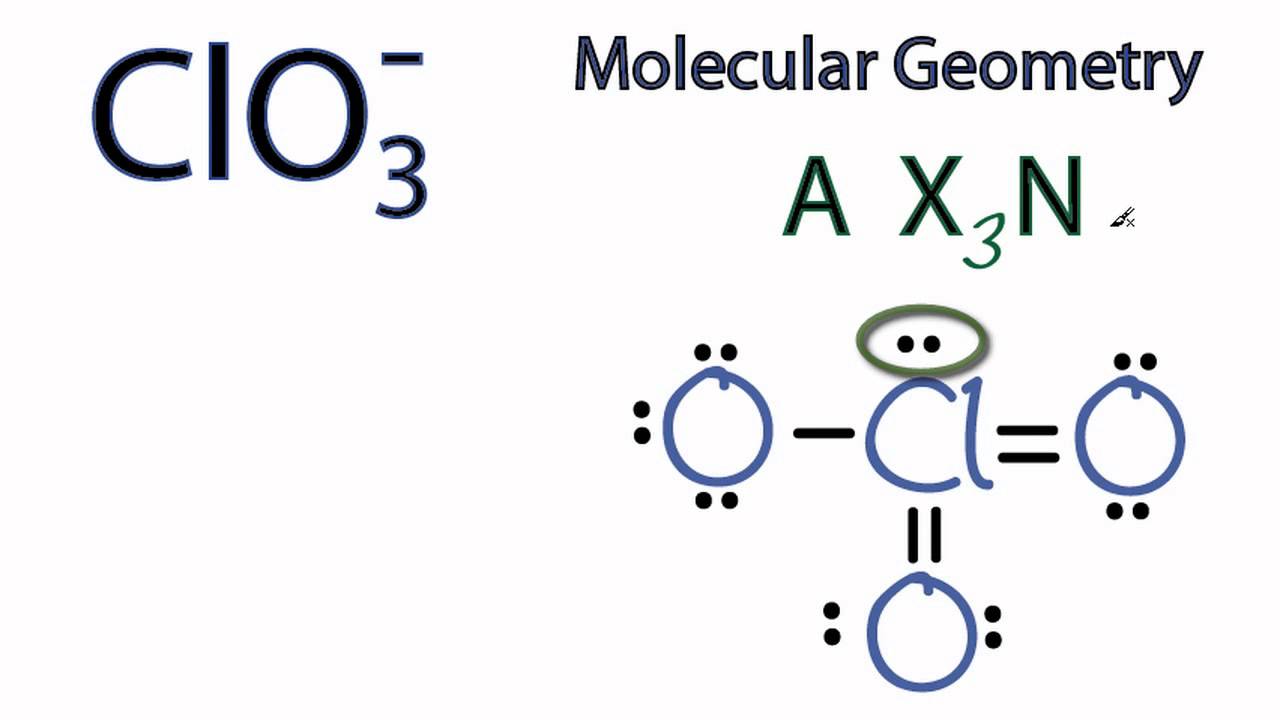
ClO3 Molecular Geometry / Shape and Bond Angles Chlorate Ion YouTube
Clo3+ Lewis Structure Bond Angle Drawing Easy

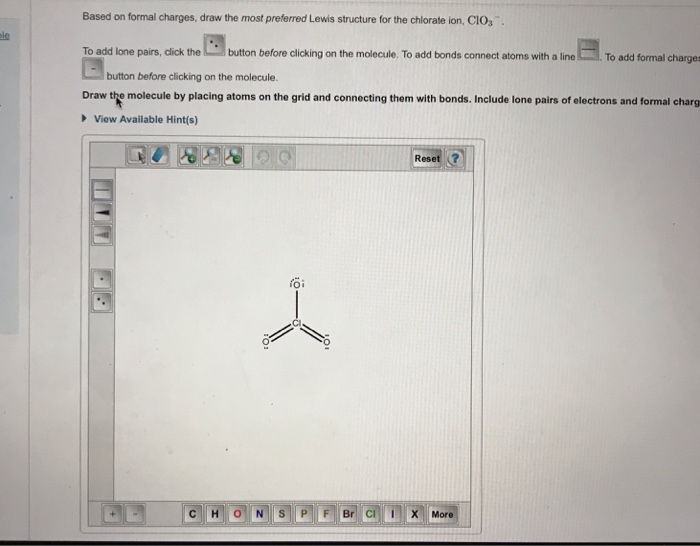
ClO3 Lewis Structure, Molecular Geometry, Hybridization, and Polarity

Clo3 Lewis Structure Molecular Geometry Hybridization And Polarity

Lewis dot structure of chlorate ion How to Draw the Lewis Structure
Put One Electron Pair In Each Bond4.
It Will Hold More Than 8 Electrons.
Web 405K Views 10 Years Ago.
70 More Lewis Dot Structures.
Related Post: