Draw The Lewis Structure Of Bf3
Draw The Lewis Structure Of Bf3 - Calculating the total valence electrons. The central b atom is sp2 hybridized. Web lewis structure of boron trifluoride (bf 3) is shown below and you can see each fluorine atom has made a single bond with boron atom. € what is the the shape (molecular geometry) of bf3? Web drawing lewis structures for molecules with one central atom: Using formal charges to determine how many bonds to make, a different perspective. What are its physical properties? Web may 28, 2022 by biswarup chandra dey. In this tutorial, we will learn how to draw the lewis structure of bf 3. This problem has been solved! Here, the given molecule is bf3. Watch the video and see if you missed any steps or information. If you're not sure you have the best lewis structure for bf 3 you can calculate the formal charges. For the bf3 structure use the periodic table to find the total number. #3 calculate and mark formal charges on the atoms, if. We should also have some understanding that what is boron trifluoride? How to draw bf3 lewis structure? This problem has been solved! If you're not sure you have the best lewis structure for bf 3 you can calculate the formal charges. Let’s discuss each step in more detail. Find the total valence electrons in bf3 molecule. Web drawing lewis structures for molecules with one central atom: Web bf3 draw the lewis structure for bf3 in the box at the right, including lone pairs. In this video, we are going to draw the lewis structure for. #1 first draw a rough sketch. Web bf3 draw the lewis structure for bf3 in the box at the right, including lone pairs. The bf 3 lewis structure is similar to bcl 3 and bbr 3 since cl and br are in group 7 and have 7 valence electrons. If you're not sure you have the best lewis structure for bf 3 you can calculate the. Drawing lewis structures for bf3, pf3 and brf3; Web following the octet rule for lewis dot structures leads to the most accurate depictions of stable molecular and atomic structures and because of this we always want to use the octet rule when drawing lewis dot structures. A prerequisite for accurately portraying the bf3 lewis structure is determining the cumulative valence. How to draw bf3 lewis structure? For the central boron atom: #3 calculate and mark formal charges on the atoms, if required. If you're not sure you have the best lewis structure for bf 3 you can calculate the formal charges. We should also have some understanding that what is boron trifluoride? Web in simple words, we can say that drawing lewis’s structure is necessary to explain all the structural properties including bond angle, hybridization, geometry, etc. Web draw the lewis structure for bf3 in the window below and then answer the questions that follow. Web following the octet rule for lewis dot structures leads to the most accurate depictions of stable. Web may 28, 2022 by biswarup chandra dey. Web use these steps to correctly draw the bf 3 lewis structure: By the end of this section, you will be able to: #2 mark lone pairs on the atoms. Watch the video and see if you missed any steps or information. What are its physical properties? #3 calculate and mark formal charges on the atoms, if required. Drawing lewis structures for bf3, pf3 and brf3; Try to draw the bf 3 lewis structure before watching the video. Web may 28, 2022 by biswarup chandra dey. Web following the octet rule for lewis dot structures leads to the most accurate depictions of stable molecular and atomic structures and because of this we always want to use the octet rule when drawing lewis dot structures. The bf 3 lewis structure is similar to bcl 3 and bbr 3 since cl and br are in group 7 and. Web drawing lewis structures for molecules with one central atom: Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. • do not include overall ion charges or formal charges in your drawing. Drawing lewis structures for bf3, pf3 and brf3; Web may 28, 2022 by biswarup chandra dey. Steps of drawing bf3 lewis structure. Web in simple words, we can say that drawing lewis’s structure is necessary to explain all the structural properties including bond angle, hybridization, geometry, etc. #1 first draw a rough sketch. This shape makes an equilateral triangle with each side making a. Web bf3 draw the lewis structure for bf3 in the box at the right, including lone pairs. For the central boron atom: Try structures similar to bf 3 for more practice. Boron (b) doesn't need 8 valence electrons to have an octet (boron often only needs 6). The number of lone pairs = the number of single bonds = the number of double bonds = 2. The central boron atom a. Calculate the total number of valence electrons.
Bf3 Lewis Structure Lone Pairs Draw Easy
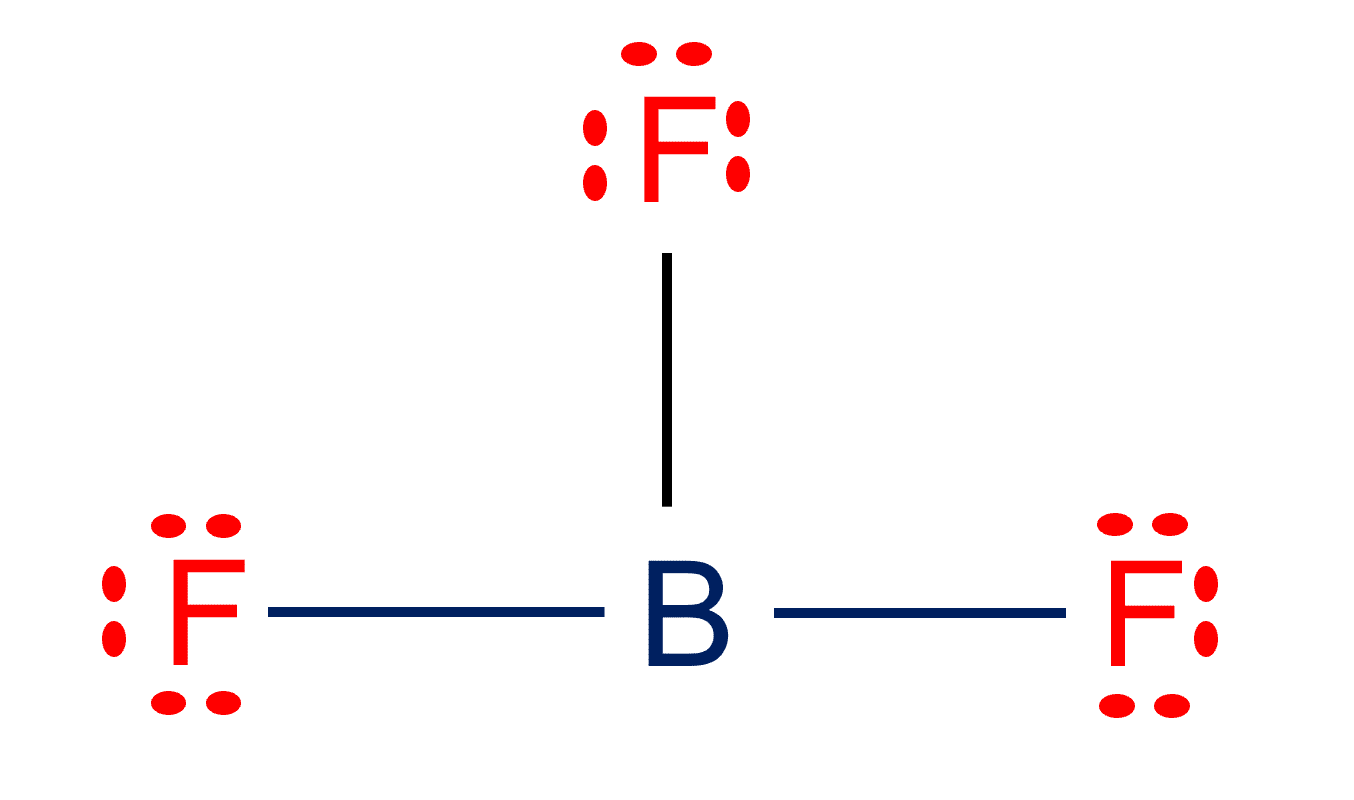
BF3 Lewis structure Molecular geometry, Hybridization, and Polarity

BF3 Lewis Structure (Boron Trifluoride) YouTube

BF3 Lewis Structure, Molecular Geometry, and Hybridization

How to draw BF3 Lewis Structure? 5

How to Draw Lewis Dot Structure of BF3 YouTube

BF3 Lewis Structure How to Draw the Lewis Structure for BF3 YouTube
Molecular Geometry CK12 Foundation

How to Draw the Lewis Dot Structure for BF3 Boron trifluoride YouTube

BF3 lewis structure and Molecular Geometry YouTube
In Order To Draw The Lewis Structure Of Bf3, First Of All You Have To Find The Total Number Of Valence Electrons Present In The Bf3 Molecule.
100% (8 Ratings) Share Share.
The Central B Atom Is Sp2 Hybridized.
Web 317K Views 10 Years Ago.
Related Post:
