Draw The Lewis Structure For Bcl3
Draw The Lewis Structure For Bcl3 - The number of lone pairs the number of single bonds = the number of double bonds = 2. (can be more then one) a) dispersion. The bcl 3 lewis structure is similar to bf 3 and bbr 3 since f and br are in group 7 and have 7 valence electrons. Web steps of drawing bcl3 lewis structure. Lewis structures show all of the valence electrons in an atom or molecule. Since there are 3 chlorines, the total is \ (3 + 3 \times 7 = 24\) valence electrons. Draw lewis structures depicting the bonding in simple molecules. In order to draw the lewis structure of bcl3, first of all you have to find the total number of valence electrons present in the bcl3 molecule. With three chlorine atoms in bcl3, the total valence electrons sum up to 3 (from boron) + 7 x 3 (from chlorine) = 24 valence. Web welcome back to our channel and in today's video, we will help you determine the lewis structure of boron trichloride. Write lewis symbols for neutral atoms and ions. First, determine the total number of valence. Count the total number of valence electrons. Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Part b determine molecular geometry of bcl3. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web draw lewis structures depicting the bonding in simple molecules. We also use lewis symbols to indicate the formation of covalent bonds, which are shown in lewis structures, drawings that describe the bonding in molecules and polyatomic ions. 138k views 10 years ago. The. Now, let’s take a closer look at each step mentioned above. Boron (b) doesn't need 8 valence electrons to have an octet (boron often only needs 6). Web welcome back to our channel and in today's video, we will help you determine the lewis structure of boron trichloride. For the central boron atom: In order to draw the lewis structure. Find the total valence electrons in bcl3 molecule. (can be more then one) a) dispersion. Web to draw the lewis structure for bcl3, follow these steps: #1 draw a rough skeleton structure. A) calculate the total number of valence electrons in bcl3: Boron (b) has 3 valence electrons, and each chlorine (cl) atom possesses 7 valence electrons. 138k views 10 years ago. Since there are 3 chlorines, the total is \ (3 + 3 \times 7 = 24\) valence electrons. Draw lewis structures depicting the bonding in simple molecules. Find the total valence electrons in bcl3 molecule. I also go over formal charge, hybridization, shape and bond angle. Drawing lewis dot structures for bcl3. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Web 5 steps to draw the lewis structure. A)linear b)bent c)trigonal planar d)trigonal pyramidal part c what kind of intermolecular forces are present in bcl3? Write lewis symbols for neutral atoms and ions. There are a few exceptions to this rule, with boron being one of them. Web drawing the lewis structure for bcl 3. #4 calculate formal charge and check stability (if octet is already completed on. Drawing lewis dot structures for bcl3. Web 5 steps to draw the lewis structure of bcl3. Web to draw lewis structures for molecules and polyatomic ions with one central atom. Boron (b) has 3 valence electrons, and each chlorine (cl) has 7 valence electrons. Draw lewis structures depicting the bonding in simple molecules. #1 draw a rough skeleton structure. Thus far, we have discussed the various types of bonds that form between atoms and/or ions. Web 5 steps to draw the lewis structure of bcl3. Web welcome back to our channel and in today's video, we will help you determine the lewis structure of boron trichloride. A)linear b)bent c)trigonal planar d)trigonal pyramidal part. #4 calculate formal charge and check stability (if octet is already completed on central atom) let’s one by one discuss each step in detail. Web draw lewis structures depicting the bonding in simple molecules. In all cases, these bonds involve the sharing or transfer of valence shell electrons between atoms. Thus far in this chapter, we have discussed the various. Web lewis structures are drawn in accordance with the octet rule, where each atom in the molecule tries to obtain 8 electrons in its outermost shell. In order to draw the lewis structure of bcl3, first of all you have to find the total number of valence electrons present in the bcl3 molecule. Web i quickly take you through how to draw the lewis structure of bcl3, (boron trichloride). Here, the given molecule is bcl3. A lewis structure is a way to show how atoms share electrons when they form a molecule. Web steps of drawing bcl3 lewis structure. Count the total number of valence electrons. A)linear b)bent c)trigonal planar d)trigonal pyramidal part c what kind of intermolecular forces are present in bcl3? With three chlorine atoms in bcl3, the total valence electrons sum up to 3 (from boron) + 7 x 3 (from chlorine) = 24 valence. The number of lone pairs the number of single bonds = the number of double bonds = 2. Boron (b) has 3 valence electrons, and each chlorine (cl) has 7 valence electrons. This video tutorial will explain how to draw the lewis dot structure and molecular geometry for boron trichloride (bcl3). Calculate the total number of valence electrons. A) calculate the total number of valence electrons in bcl3: Web to draw the lewis structure for bcl3, follow these steps: #3 if needed, mention formal charges on the atoms.
Bcl3 Lewis Structure / How Many Lone Pairs Are On The Central Ato

Lewis dot structure of BCl3 Boron trichloride lewis structure

Bcl3 Lewis Structure How To Draw The Lewis Structure For Bcl3 Otosection
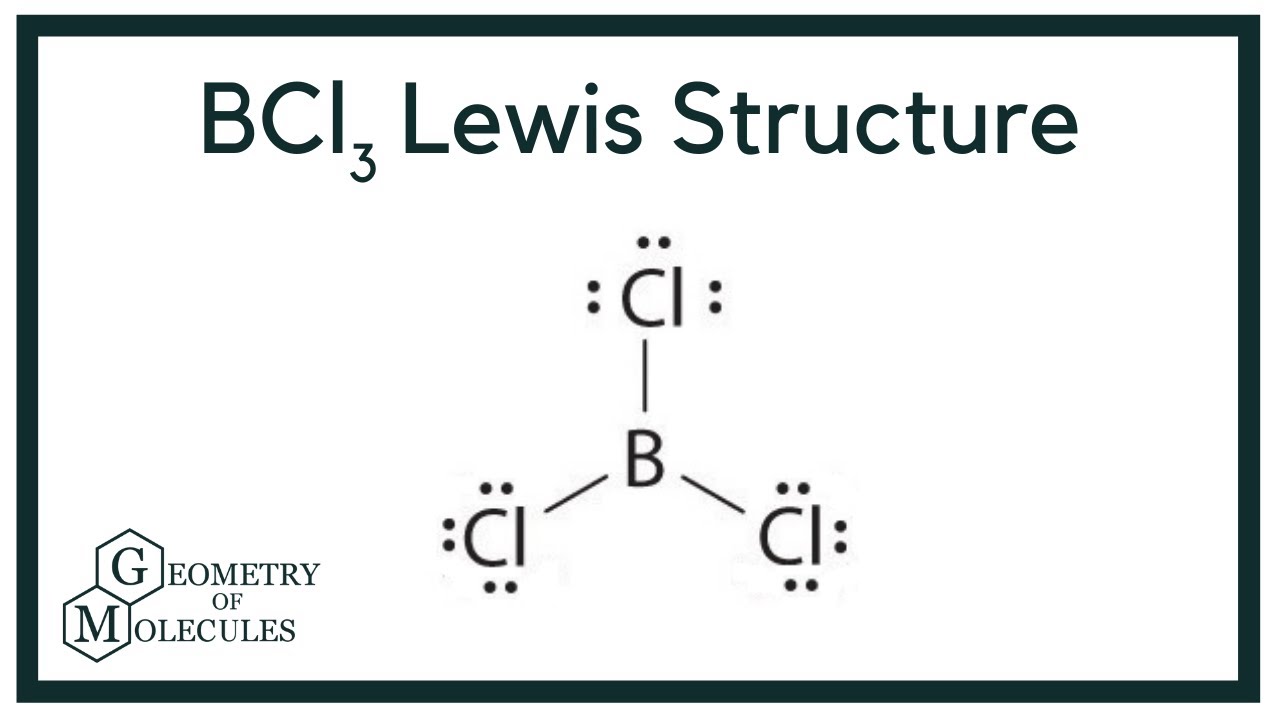
BCl3 Lewis Structure (Boron Trichloride) YouTube

Draw the Lewis structure of boron trichloride BCl3 YouTube

Draw the Lewis dot structure for boron trichloride, BCl_3. Quizlet

Bcl3 Lewis Structure Molecular Geometry And Hybridization itechguide

BCl3 Lewis Structure in four simple steps What's Insight

BCl3 Lewis Structure How to Draw the Lewis Structure for BCl3 YouTube

draw the lewis structure for bcl3 in the marvin window below and then
Bcl3 Draw The Lewis Dot Structure For Bcl3.
Web A Video Explanation Of How To Draw The Lewis Dot Structure For Boron Trichloride, Along With Information About The Compound Including Formal Charges, Polarity, Hybrid Orbitals, Shape, And Bond.
Find The Total Valence Electrons In Bcl3 Molecule.
Boron (B) Doesn't Need 8 Valence Electrons To Have An Octet (Boron Often Only Needs 6).
Related Post: