Draw A Picture Of An S Orbital
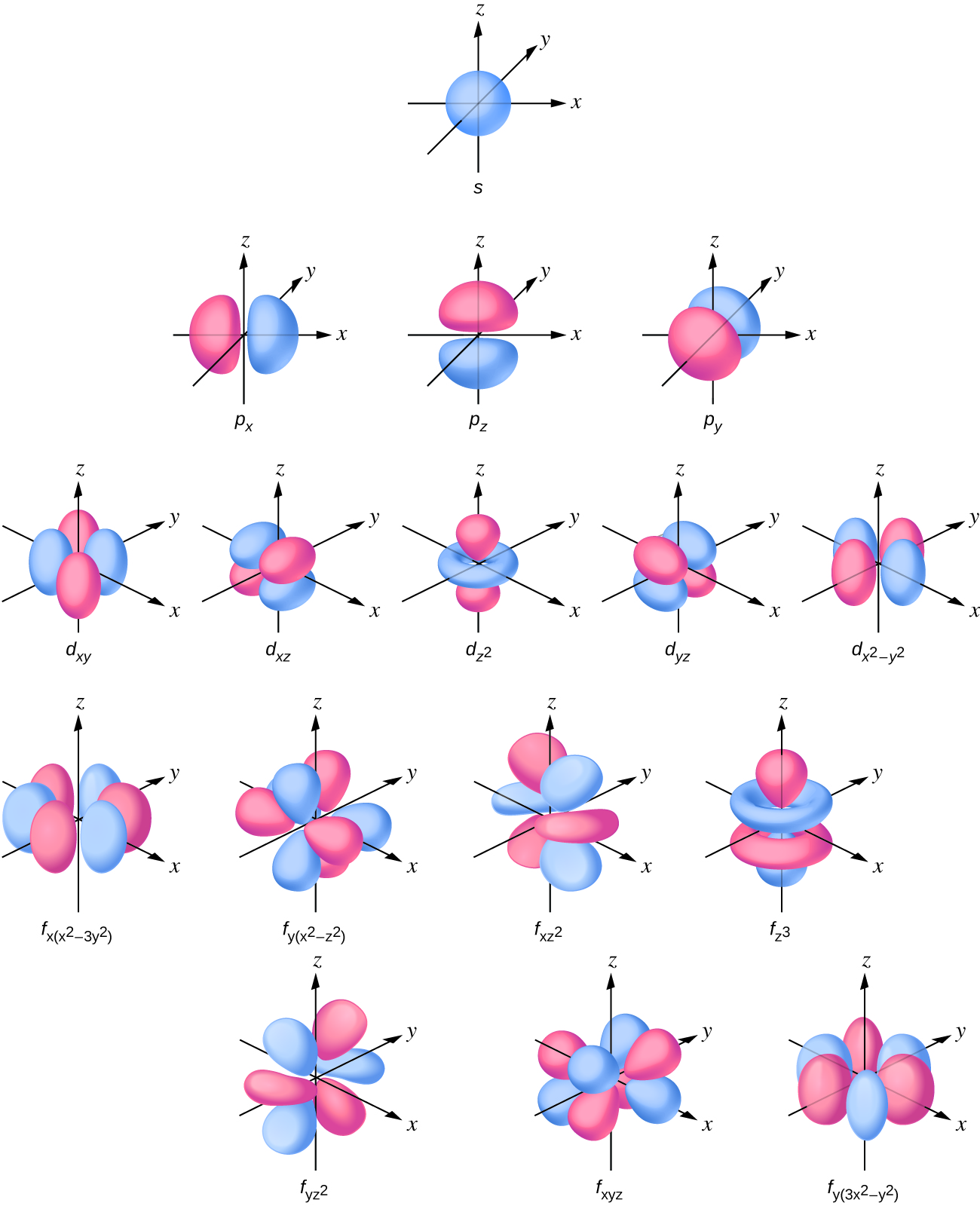
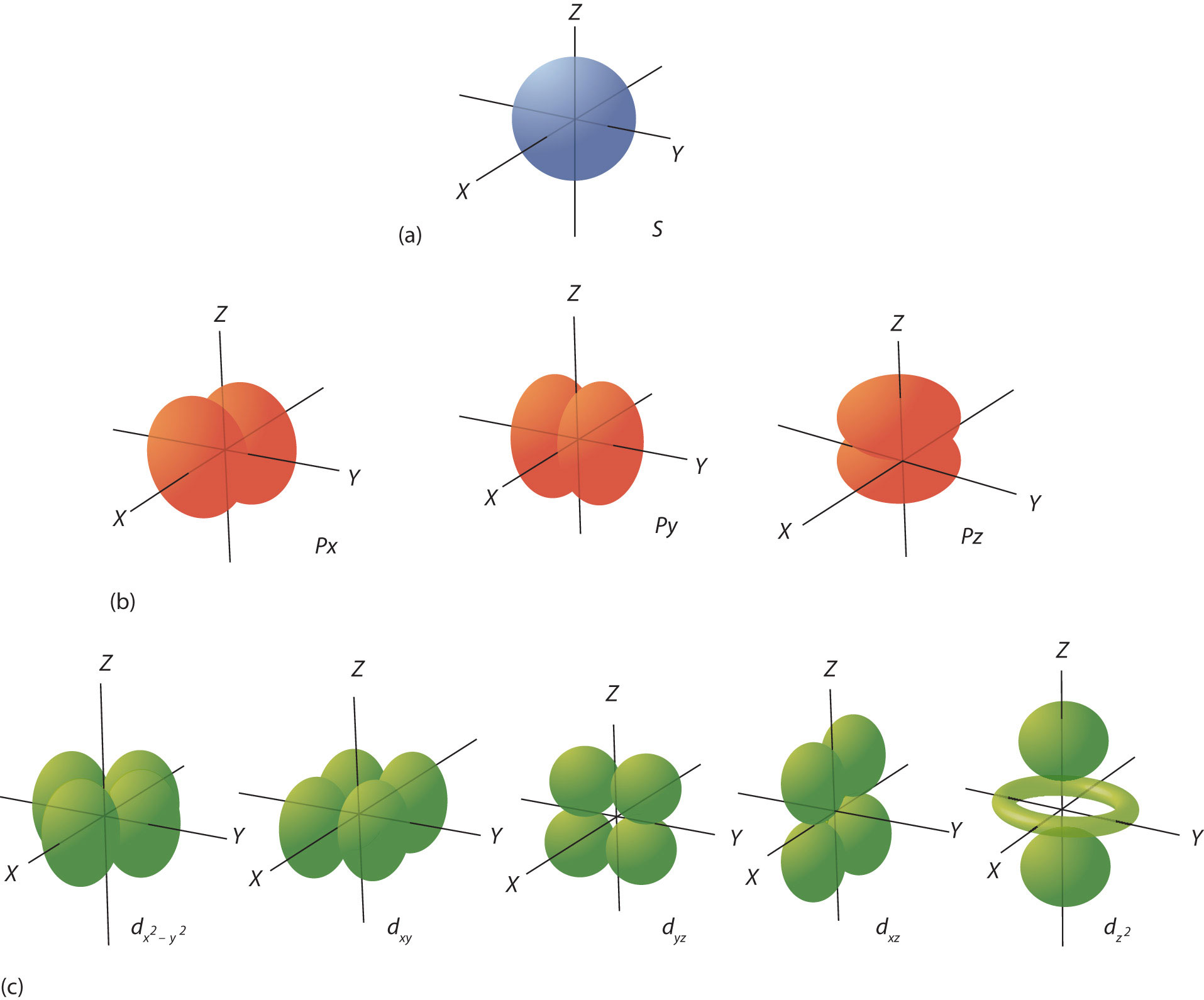
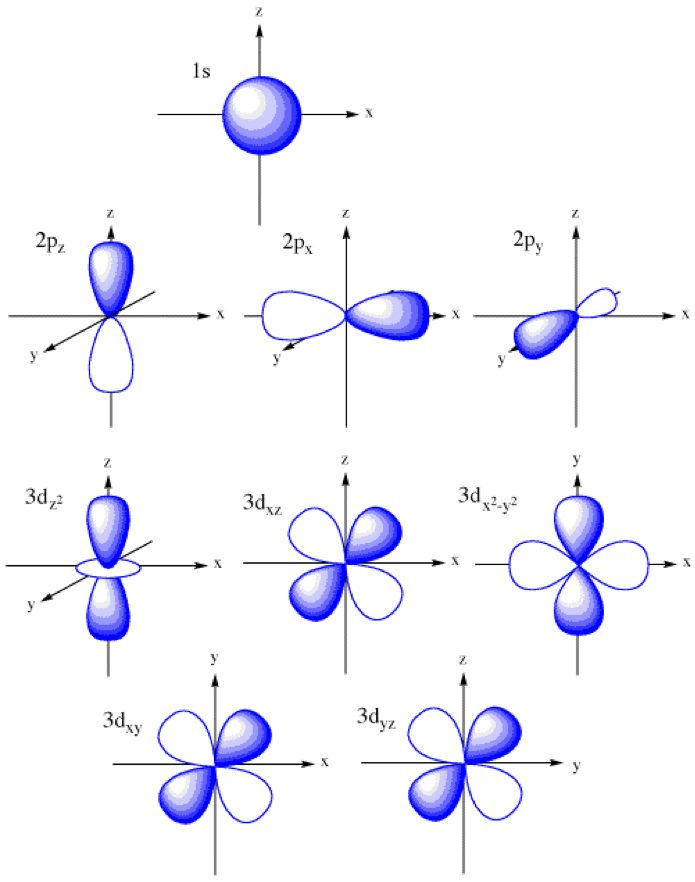
Draw A Picture Of An S Orbital - Web the letter s indicates the shape of the orbital: Sketch the shapes of s and p orbitals. In a typical drawing of orbital, we first plot the radial wave function and the angular part is superimposed. S orbitals are spherically symmetric around the nucleus— they look like hollow balls made of chunky material with the nucleus at the center. Describe the physical significance of an orbital. Next, draw the first orbital around the nucleus. Make certain that you can define, and use in context, the key terms below. For a p orbital, draw a figure eight; The subscript x,y and z referrers to the axis the orbital is on in 3d. An orbital is a space where a specific pair of electrons can be found. Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. The fifth d orbital is shaped like an elongated dumbbell with a doughnut around its middle. Sketch the shapes of s and p orbitals. Next, draw the first orbital around the nucleus. The. They are at a slightly higher level than the 4s. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web for the s orbitals, first draw an arrow pointing up and an arrow pointing down. After completing this section, you should be able to. Imagine shells around the nucleus, that get bigger and. Now, draw the second orbital. This problem has been solved! Web the letter s indicates the shape of the orbital: This process is the same for the d and f orbitals. List the atomic orbitals from 1 s to 3 d in order of increasing energy. For the p orbitals, draw one arrow pointing up on each of the lines first. Web the impossibility of drawing orbits for electrons. We classified the different orbital into shells and sub shells to distinguish them more easily. The shapes of some typical orbitals are discussed below. Imagine shells around the nucleus, that get bigger and bigger. 2) looking at our cheat sheet, draw the orbitals one at a time, adding electrons as you go, until you reach a total of 16 electrons. Let's learn about the shapes of atomic orbitals in this video lesson.1. Next, draw the first orbital around the nucleus. An orbital is a space where a specific pair of electrons can be found.. A p orbital consists of two lobes of electron density on either side of the nucleus. Web a p orbital along the y axis is labeled p y and one along the z axis is a p z orbital. This problem has been solved! An s orbital is a sphere. For the p orbitals, draw one arrow pointing up on. In a typical drawing of orbital, we first plot the radial wave function and the angular part is superimposed. 6.6k views 3 years ago drawing figures. Repeat step 3 until all of the orbitals have been drawn. This is also due to the history when they were discovered. An orbital is a space where a specific pair of electrons can. The orbitals in an atom are organized into different layers or electron shells. Make sure you check here first.what quantum numbers can apply to electrons in s orbitals? We classified the different orbital into shells and sub shells to distinguish them more easily. Web the letter s indicates the shape of the orbital: S orbitalsthere is only 1 s orbital. Sketch the shapes of s and p orbitals. Web depict the orbital shapes are intended to describe the region encompassing 90−95% probability density. An orbital is a space where a specific pair of electrons can be found. 6.6k views 3 years ago drawing figures. S orbitals are spherically symmetric around the nucleus— they look like hollow balls made of chunky. Web depict the orbital shapes are intended to describe the region encompassing 90−95% probability density. Web in sp hybridization, one s orbital and one p orbital hybridize to form two sp orbitals, each consisting of 50% s character and 50% p character. In molecular orbital theory, the bonding between atoms. 2) looking at our cheat sheet, draw the orbitals one. Web the molecular orbital theory, initially developed by robert s. Web the impossibility of drawing orbits for electrons. 2) looking at our cheat sheet, draw the orbitals one at a time, adding electrons as you go, until you reach a total of 16 electrons. O what quantum numbers can apply to electrons in sorbitals? To do this, simply draw a line around the nucleus. S orbitalsthere is only 1 s orbital in each s subshell.shape of s. Web 1) look at the periodic table to see how many electrons sulfur has. For the p orbitals, draw one arrow pointing up on each of the lines first. List the atomic orbitals from 1 s to 3 d in order of increasing energy. This problem has been solved! This type of hybridization is required whenever an atom is surrounded by four groups of electrons. In molecular orbital theory, the bonding between atoms. Web depict the orbital shapes are intended to describe the region encompassing 90−95% probability density. The subscript x,y and z referrers to the axis the orbital is on in 3d. Notice that the s orbital always has a slightly lower energy than the p orbitals at the same energy level, so the s orbital always fills with electrons before the corresponding p orbitals. Below are dot density diagrams, boundary surface diagrams, and a rotating image.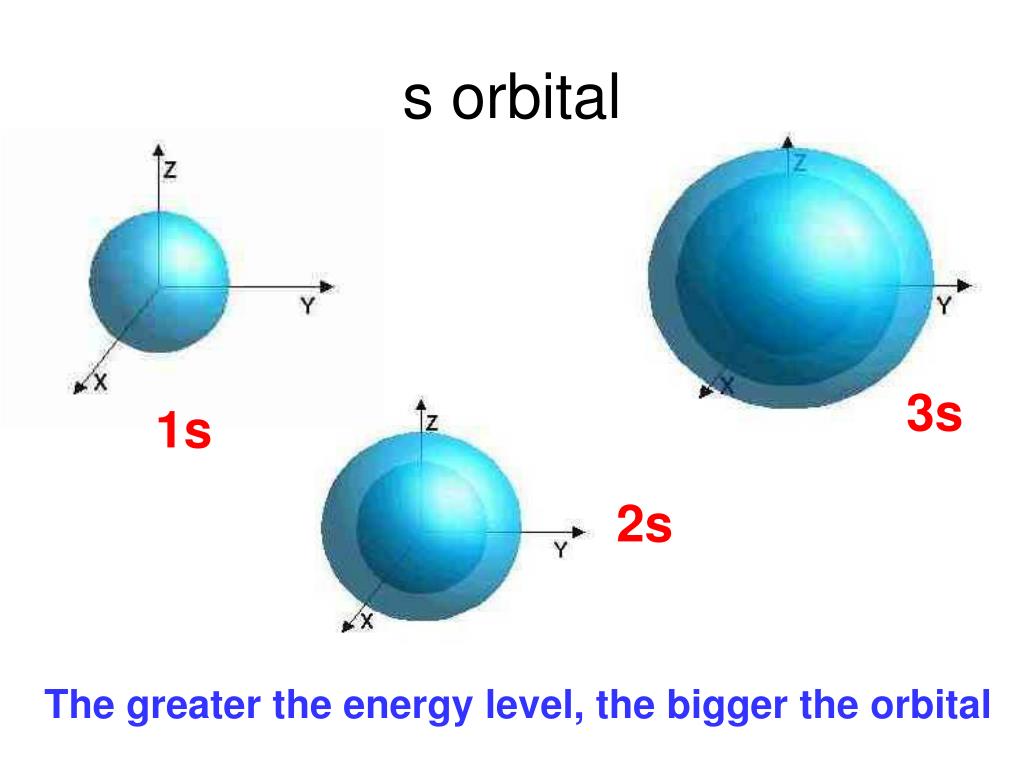
PPT Chapter 5 Electrons In Atoms PowerPoint Presentation, free

Atoms and Atomic Structure HubPages
8.3 Development of Quantum Theory CHEM 1114 Introduction to Chemistry

Biochemistry Glossary Orbitals 2. Shape Draw It to Know It
3.7 Electron Arrangement The Quantum Model Chemistry LibreTexts
Atomic orbitals explained polizhuge
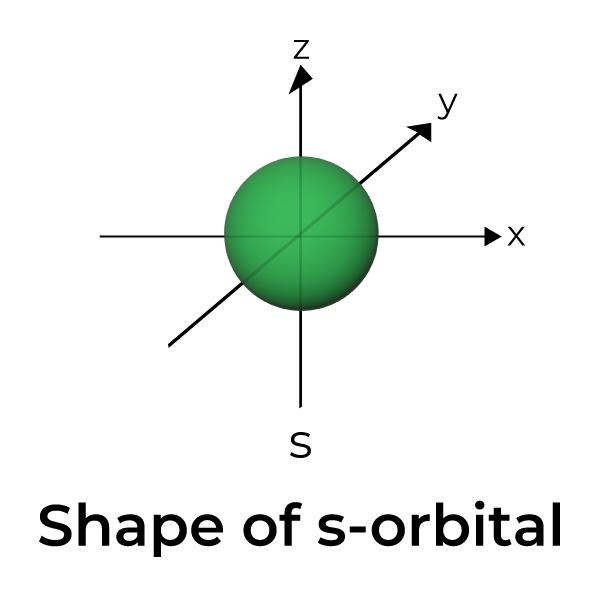
Shapes of Atomic Orbitals Shape of s, p, d, f Orbitals, FAQs, Examples

Shapes of Atomic Orbitals — Overview & Examples Expii

12.1.5 Draw the shape of an s orbital and the shapes of the p x , p y
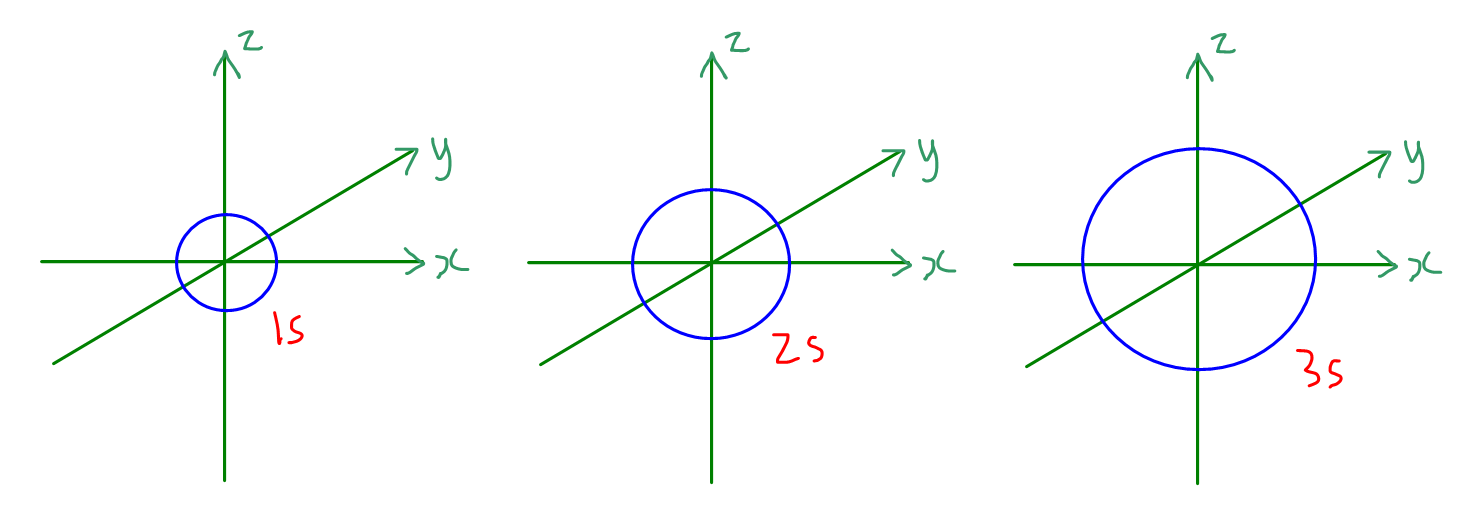
How to Draw Shapes of Orbitals
After Completing This Section, You Should Be Able To.
Make Certain That You Can Define, And Use In Context, The Key Terms Below.
For An F Orbital, See Below.
Web For The S Orbitals, First Draw An Arrow Pointing Up And An Arrow Pointing Down.
Related Post: